Stifled Cells: How Zika Halts the Developing Brain
- Published23 Feb 2017
- Reviewed23 Feb 2017
- Author Teal Burrell
- Source BrainFacts/SfN
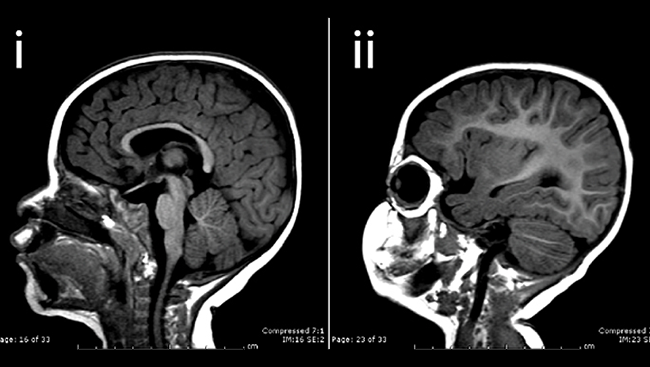
In 2015, health officials in Latin America noticed an alarming trend: as reported cases of Zika virus infection spiked so did the number of babies born with microcephaly — a rare and serious birth defect resulting in a shrunken brain and abnormally small head. The apparent link between Zika and birth defects led the World Health Organization to declare the mosquito-borne virus a global health emergency in early 2016.
As the Zika virus threatens to spread around the world, fears of its devastating effects have outpaced it. Even so, how the virus inflicts its damage on the developing brain remains a mystery. As neuroscientists race to figure out what exactly happens inside infected brain cells and how the cells might fight back, they are laying the groundwork to develop new drugs and treatments.
Birth Rates Down, Death Rates Up
Adults infected with Zika may develop some mild symptoms like fever, rash, joint, and muscle pain, and pink eye. Most have few — if any — symptoms, and many never realize they’ve been infected at all. But for pregnant women, Zika infection — even a silent one — carries more serious risks: the virus can pass through the placenta and harm the developing fetus, potentially causing miscarriage, stillbirth, or severe developmental problems such as microcephaly. Babies with microcephaly may have trouble feeding in addition to developmental delays in speech and motor skills. They may have seizures, intellectual disabilities, and hearing and vision problems. In severe cases, it can be life threatening.
Cells in the massively growing fetal brain multiply at exponential rates: one cell splits into two; two splits into four and so on creating ever more cells. Somehow, Zika infection halts this process — rather than one brain cell splitting in two and creating more cells, many infected brain cells simply stop dividing, while others die off. With fewer cells being born and more cells dying, the brain can’t grow to its normal size.
The question is how? Patricia Garcez and a team of researchers at Federal University of Rio de Janeiro speculated that Zika tampered with the genes instructing cells to divide and mature, so they injected human stem cells — cells with the ability to turn into any of the major cell types in the brain — with a strain of the virus and identified what genes were turned on or off. In infected stem cells, Zika virus disrupts more than five hundred genes. Healthy, uninfected cells employ a slew of genes to make cells multiply and instruct them to assume certain roles. But the Zika virus switched these genes off, meaning they couldn’t provide the instructions young brain cells need to grow and assemble into a normally functioning brain.
While these findings captured the effects of Zika infection on gene activity in the individual cells, they don’t address how a developing brain copes. Studying a fetal brain as it develops is impossible, so researchers have turned to “mini-brains” or organoids for answers. When grown in liquid rather than attached to a lab dish, stem cells develop into three-dimensional, floating balls organized like the fetal brain, with distinct layers of different brain cell types. Xuyu Qian and colleagues at Johns Hopkins University found the virus put a stop to the cells dividing, while also killing existing cells. And infected organoids had smaller layers of neurons and were smaller overall as a result, suggesting that neurons are particularly vulnerable and that it is their depletion specifically that leads to the smaller brain and head size. What’s more, infected cells turn into virus factories, producing more and more virus particles capable of infecting neighboring cells and spreading devastation throughout the nascent brain.
The Brain Fights Back
But the fetal brain doesn’t take this lying down; it actively fights to prevent Zika from inflicting too much damage.
While Zika shuts off genes needed to make new neurons, one study found that genes for making another type of brain cell — astrocytes — were turned up. Astrocytes nourish neurons, fight infections, and help repair the nervous system after damage. The study, which used mouse stem cells, also found that infected cells had more immune proteins boosting astrocytes’ ability to fight infection.
“It gives us an idea as to how the neural stem cells are responding to the virus and how they’re attempting to combat the Zika virus,” says Olivia Lossia, of Central Michigan University who conducted the study. “There’s kind of a fight going on between the virus and the cells — the virus is trying to further the infection while the cells are obviously trying to protect against it.”
Those defensive efforts may cause more harm than good. A recent study showed that human neural stem cells infected with Zika virus stopped producing new cells because a protein vital to the process took on a new job. Instead of regulating cell division, the protein assumed immune response duties, trying to fight the virus. By forsaking its role in creating new cells, the reassigned protein may play a role in the virus’s devastation.
Separating the Vulnerable from the Resilient
Still, the fetal brain sometimes mounts a successful defense. “Only a subset of infants born to Zika-infected mothers actually exhibit neurological deficits such as microcephaly,” says Erica McGrath of the University of Texas Medical Branch. A study from last July estimated between 1 and 13 percent of babies born to mothers infected in the first trimester developed microcephaly.
McGrath grew stem cells from three different fetal brains in the lab and infected them with Zika. Infection killed cells and hindered cell birth in two of the stem cell lines — but not in the third. It had a normal number of cells. In the more vulnerable cell lines, infection amplified expression of genes promoting both the immune response and cell death, and diminished expression of genes encouraging cell survival. The resilient cell line, on the other hand, turned up expression of cell survival genes.
This suggests there is variation in the genetic response to Zika in different individuals, making some people more susceptible to the disease. “We are hoping our findings will pave the way to identifying high risk individuals that might be at a greater risk than the rest of the population for developing neurological deficits,” says McGrath.
Understanding the reasons behind susceptibility and resiliency, along with how Zika influences genes important for cell birth, death, and fighting infection, may help scientists develop successful treatments. By knowing more about the people and genes most vulnerable to Zika’s assault, researchers might be able to protect them both.
CONTENT PROVIDED BY
BrainFacts/SfN
References
Garcez P. Zika virus hijacks genetic program that signals developing cells to multiple, specialize. Press conference at the annual meeting of the Society for Neuroscience (2016).
Johansson M. Zika and the risk of microcephaly. The New England Journal of Medicine. 375: 1-4 (2016).
Lossia O. Study suggests Zika infection may spur production of astrocytes. Press conference at the annual meeting of the Society for Neuroscience (2016).
Mcgrath E. Sorting out why some babies’ brains are more susceptible to Zika than others. Press conference at the annual meeting of the Society for Neuroscience (2016).
Onorati M, Zhen L, Fuchen L, et al. Zika virus disrupts phospho-TBK1 localization and mitosis in human neuroepithelial stem cells and radial glia. Cell Reports. 16: 2576-2592 (2016).
Qian X. Lab-made ‘mini-brains’ allow researchers to study Zika’s effects on developing brains. Press conference at the annual meeting of the Society for Neuroscience (2016).
Zika. Centers for Disease Control and Prevention. February 16, 2017.