Scientists Created a Color-Coded Atlas of the Worm Nervous System
- Published22 Dec 2021
- Author Alexis Wnuk
- Source BrainFacts/SfN
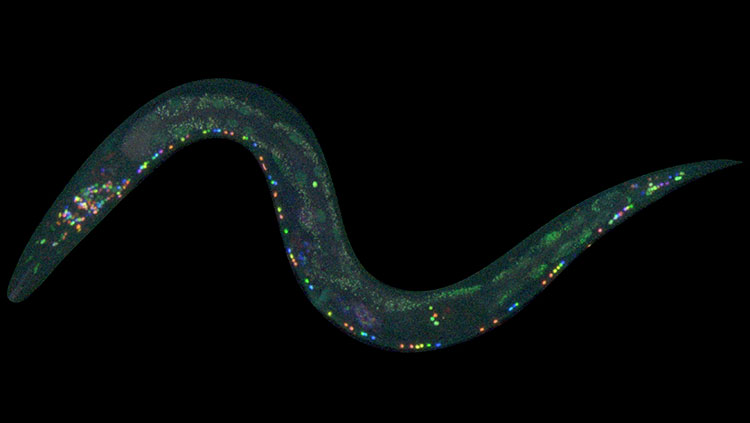
For neuroscientists, the beauty of the C. elegans nematode lies in its simplicity: just 302 neurons comprise the tiny worm’s nervous system. Still, it can be hard to distinguish one type of a neuron from another. Or to know for sure that a neuron in one worm’s tail is identical to the tail neuron in another worm.
Earlier this year, scientists unveiled a new technique to color code the 302 neurons in a living worm’s nervous system. Called NeuroPAL — short for a neuronal polychromatic atlas of landmarks — the technique makes it possible to identify neurons based on their location and color marker.
Neuroscientists can visualize neurons under the microscope using packages of genetic material and fluorescent proteins called reporter genes. Reporter genes target specific types of cells, tagging them with fluorescent proteins and making them identifiable.
But a nonspecific reporter might tag many different kinds of neurons, obscuring the unique features of each. And using multiple reporters is a long and tedious process. So, the researchers fused 41 different reporter genes together, each loaded with a palette of one or more fluorescent “paints.” The carefully chosen reporters targeted different types of neurons, with nearly every neuron taking up a unique combination of genetic coloring instructions and glowing a different color.
What’s more, the paint-by-numbers approach works the same in every C. elegans worm. That distinguishes it from the “Brainbow” neuron painting technique, where neurons randomly take up colors. With NeuroPAL, scientists can identify every type of neuron in a living worm — a first in a living animal.
Being able to ID all these neurons will make it easier to figure out which neurons are involved in different behaviors. To that end, the research team used their NeuroPAL worms to study how the neurons communicate with each other. Using a separate reporter gene that made calcium ions fluoresce, they visualized calcium flooding into the neurons, signaling neuronal activity. The results were surprising: even simple behaviors like smelling and tasting set off a complex symphony of neuronal activity.
Maybe the simple C. elegans isn’t so simple after all.
CONTENT PROVIDED BY
BrainFacts/SfN
References
Taylor, S. R., Santpere, G., Weinreb, A., Barrett, A., Reilly, M. B., Xu, C., Varol, E., Oikonomou, P., Glenwinkel, L., McWhirter, R., Poff, A., Basavaraju, M., Rafi, I., Yemini, E., Cook, S. J., Abrams, A., Vidal, B., Cros, C., Tavazoie, S., … Miller, D. M. (2021). Molecular topography of an entire nervous system. Cell, 184(16), 4329-4347.e23. https://doi.org/10.1016/j.cell.2021.06.023
Yemini, E., Lin, A., Nejatbakhsh, A., Varol, E., Sun, R., Mena, G. E., Samuel, A. D. T., Paninski, L., Venkatachalam, V., & Hobert, O. (2021). NeuroPAL: A Multicolor Atlas for Whole-Brain Neuronal Identification in C. elegans. Cell, 184(1), 272-288.e11. https://doi.org/10.1016/j.cell.2020.12.012