The brain stores memories by changing how neurons talk to each other. When one neuron fires an actional potential, another neuron activates. Over time, this connection gets stronger. Scientists can watch this play out in real time by stimulating and recording slices of brain tissue.
This is a video placed first in the 2021 Brain Awareness Video Contest.
Created by Tanja Fuchsberger
CONTENT PROVIDED BY
BrainFacts/SfN
Transcript
The capacity of the brain to learn new information and remember experiences is essential to our existence. Our memory connects our past to the present and prepares for us for the future. It helps animals and humans to adapt to the environment. Almost everything we do relies on it — finding food and knowing how to get it or remembering how to get home.
So, the question is: how does the brain store new information? Let's take a look inside!
In 1949 the Canadian psychologist Donald Hebb suggested that learning and memory occurs when two connected neurons are activated at the same time or close in time, leading to strengthening of the connection between them. This is often paraphrased as “neurons that fire together, wire together.” A neuron sends an action potential along its axon, causing it to release a neurotransmitter, which the other neuron receives at the synapse, the connection between them.
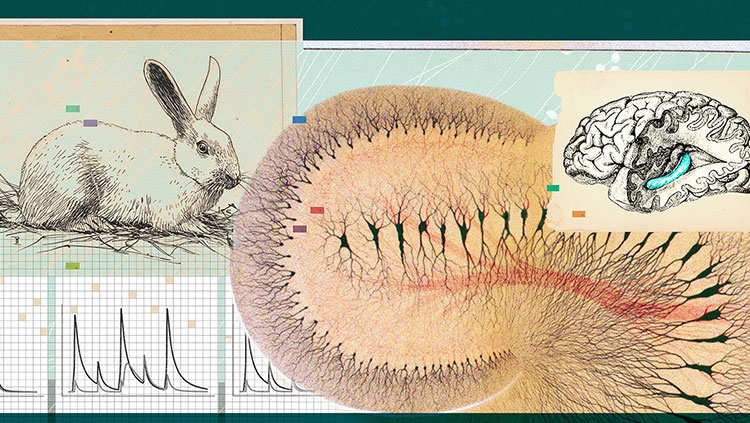
Let's do an experiment where we look at changes in synaptic strength called long term potentiation, or LTP. Here we study this in a rodent brain, but it has been observed in several different species. We prepare brain slices, and the cells in the brain tissue can be kept alive in oxygenated artificial brain fluid for several hours. We pick up a brain slice and place the tissue under a microscope. We look for an area called the hippocampus, which is known to be important for memory.
For our experiment, we place an electrode onto the slice to stimulate axons that are connected to a neuron that we will record from. This technique is called patch clamp. First, we position the stimulation electrode. Then we increase the magnification on the microscope so that we can see the cell bodies of individual neurons. We place the patch pipette on the slice. Tiny movements are controlled with a micromanipulator. We now establish a connection between the patch pipette and the membrane of the neuron at the cell body. We open the cell, and we can now monitor and control its electrical activity.
But before we start our experiment, let's take a look inside the screen. This is a 3D reconstruction of the neuron. While the patch pipette is located on the cell body, we will be recording signals that the neuron receives far out at its synapses.
Now let's start our experiment. We have placed the stimulation electrode on axons and connected it to a stimulation box. The patch pipette is connected to an amplifier, and we can now monitor the activity of the cell. We apply current and induce an action potential. On the postsynaptic cell we record the synaptic response, called excitatory postsynaptic potential, or EPSP.
Ion channels and receptors transmit the signals. The amount and location of receptors in the synapse is dynamically regulated and determines the strength of the connection. When a neuron fires an action potential, glutamate is released and opens receptors to let ions flow in. This induces the synaptic potential that we record.
To induce LTP here we will use spike-timing dependent plasticity. For this, we induce an action potential in the presynaptic neuron, and immediately after, we induce an action potential in the connected neuron. This protocol is based on Hebb's postulate. But so what does the coincident activation of the two neurons actually do? One important mechanism for LTP is that we have a coincidence detector in the synapse, the NMDA receptor. It is normally blocked by magnesium. However, when the neuron becomes activated, this block is briefly removed. If in this very moment the presynaptic neuron fires an action potential, the receptor opens and allows ions to flow in. This induces downstream signals that lead to the incorporation of more AMPA receptors in the synapse, which increases synaptic strength.
We now resume the same stimulation as before the LTP induction. And the magic has happened! The synaptic response has become larger. Synaptic changes are likely determining which cells are activated together and belong to a neuronal ensemble. So maybe this is why the cat knows that there is food in the blue ball!