Night Owl Chronotypes in a Morning Lark World
- Published21 Dec 2020
- Author Alexis Wnuk
- Source BrainFacts/SfN

Access to electricity has offered new opportunities for things at night. In doing so, it’s pushed our sleep timing back and expanded the range of individuals’ sleep preferences. But, work and school schedules haven’t really changed.
Lighting Up the Night
For two weeks in 2016, residents of two towns in Mozambique donned activity trackers. The wrist-worn devices recorded their movement, sleep, and light exposure as they went about their day. Those living in the rural village of Tengua spent long days in the fields where they grew maize and tended livestock. Others lived and worked about 20 miles away in the small market town Milange, the only locale in the larger district with access to electricity.
The residents were taking part in a study led by an international team of researchers examining how access to electricity affects sleep. Light sets our internal clocks, signaling that it’s time for activity. It stood to reason that more light at night could disrupt sleep patterns, says study author Kristen Knutson.
Indeed, Milange residents went to bed and woke up almost an hour later than people in Tengua. But they didn’t lose any sleep or sleep poorly as a result — their sleep timing simply shifted later. Access to electricity may change when we go to bed but “it doesn’t necessarily have to lead to poor sleep quality,” the Northwestern Feinberg School of Medicine’s Knutson says.
Surprisingly, it was the Tengua villagers who slept worse, likely because they slept on mats on the floor and slept with more family members to a room. To Knutson, the study highlights the challenges of examining how a single factor — access to electricity — affects sleep. “In understanding sleep patterns, you really need to consider the socio-cultural and environmental factors and context in which people are sleeping,” Knutson says.
The study offers a microcosmic glimpse of the way our environment affects our sleep — for better or worse — and how sleep patterns have changed over the course of human history.
Our circadian rhythms — the roughly 24-hour cycles of sleep and wake — are baked into our genes. Even our preferred bedtime is rooted in our DNA. In the last three decades, scientists have identified a slew of genes linked to chronotype, our individual internal clock that governs when we sleep and when we feel most alert. The Mozambique study, and others like it, show how malleable and adaptable our circadian rhythms are. Access to electricity has pushed our sleep timing back and expanded the range of individuals’ sleep preferences. But, in a world built for morning larks, night owls and even middle-of-the-road chronotypes can experience disastrous health consequences.
Nurture Over Nature
Nearly all living things — from humans to fruit flies to bacteria — run on a roughly 24-hour internal clock. Typos in the genetic code can speed up or slow down the clock; other tweaks shift it earlier or later.
In the 1970s and 80s, CalTech scientists Seymour Benzer and Ronald Konopka identified a genetic mutation in fruit flies that compressed the circadian clock. Another lengthened it. And, yet another abolished the circadian rhythm altogether. Jerry Feldman, then at SUNY Albany and now at UC Santa Cruz, found similar mutations in Neurospora crassa, a type of red bread mold.
Similar genetic variants have been found in people. In 2001, for instance, scientists at the University of Utah identified a genetic mutation responsible for a disabling circadian disruption called familial advanced sleep phase syndrome. People with this condition are extreme morning larks — they wake up around 4:30 a.m. and feel sleepy by 7 or 8 in the evening.

While circadian disruptions like these — resulting from a single, misprinted gene — offer scientists a unique opportunity to study the genetic basis of sleep timing, they are rare. For most people, the genetic picture is more muddled. Large-scale genetic studies have uncovered a host of genes linked to normal variations in sleep timing, but each likely exerts only a small effect on its own.
Rather, it seems our environment and behavior interact with our genes to drive when and how much we sleep. They also account for the tremendous variability we now see in chronotypes. “It’s nothing that we genetically or epigenetically changed in us,” says chronobiologist Till Roenneberg of the Ludwig Maximilian University of Munich.
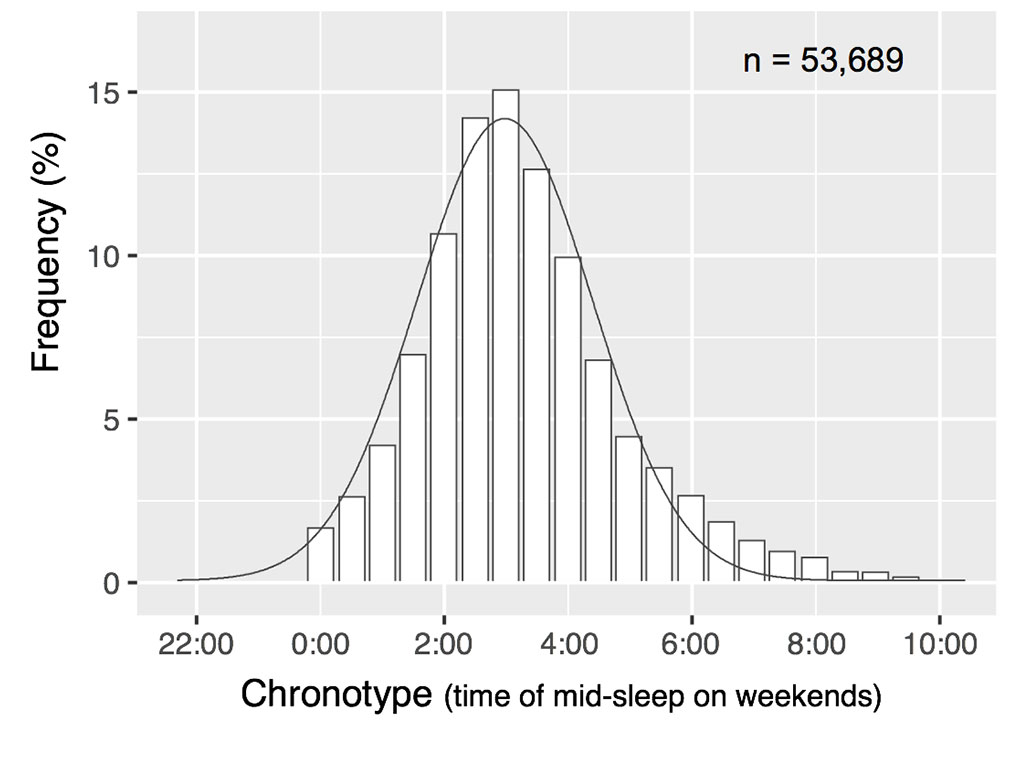
At one point in human history, the sleep timing of morning larks and night owls was probably only a few hours apart, he says. Electricity and other modern amenities have pulled the two apart. Now, a bell-curve distribution with nearly 12 hours separates the extreme morning larks from the extreme night owls but most people fall squarely in the middle.
Sleep researcher Kenneth Wright’s work best illustrates the powerful influence of the lighting environment. In 2013, the University of Colorado at Boulder neuroscientist took eight people backcountry camping in the Rocky Mountains. After one week without any electric light or electronic devices, participants’ sleep timing synchronized to the natural light-dark cycle and the differences between night owls and morning larks almost disappeared.
“When you have access to electricity, people can drift one way or the other based on their preference,” Knutson says. “We don’t go to bed the second we feel sleepy. We override that a lot more than other animals do, and so we can shape when we sleep.”
Living Against the Clock
Artificial light and electric devices have shifted our internal clocks, pushing bedtimes back and widening the spectrum of chronotypes. But our social clock, as Roenneberg calls it, has failed to keep pace: Work and social schedules favor early risers, a one-size-fits-all approach that hurts everyone else.
Evening chronotypes experience higher rates of obesity, diabetes, metabolic syndrome, and depression. They’re also more likely to smoke, use drugs, and eat poor diets. And, in a 2018 study of more than 430,000 people, Knutson and her University of Surrey collaborator, Malcolm von Schantz, found evening chronotypes had an increased risk of death during the study period compared to morning types.
Knutson doesn’t take this to mean that being an evening chronotype is inherently dangerous, however. Rather, it’s that they’re fighting against their natural biological rhythms to meet society’s expectations. To put it simply, “it’s being a night owl in a morning lark world.”
As we spend less time outside during the day and more time in front of screens at night, more of us fall into the night owl camp. In a 2012 study, Roenneberg analyzed sleep surveys collected over nine years and found the average chronotype shifted about 15 minutes later, a small but statistically significant difference. “We’ve created an environment that forces the [body] clock to be late,” Roenneberg says.
As we cut our sleep short during the work week, we accumulate sleep debt. Sleeping in on the weekends erases the debt but reverts our circadian rhythms to their natural set point. Come Monday, we have to reset — a phenomenon Roenneberg calls “social jetlag.”
Maintaining a consistent sleep schedule by waking up earlier on the weekends fails to solve the problem. “It’s not sleeping in on the weekend but getting up too early during the week that makes us sick,” he says.
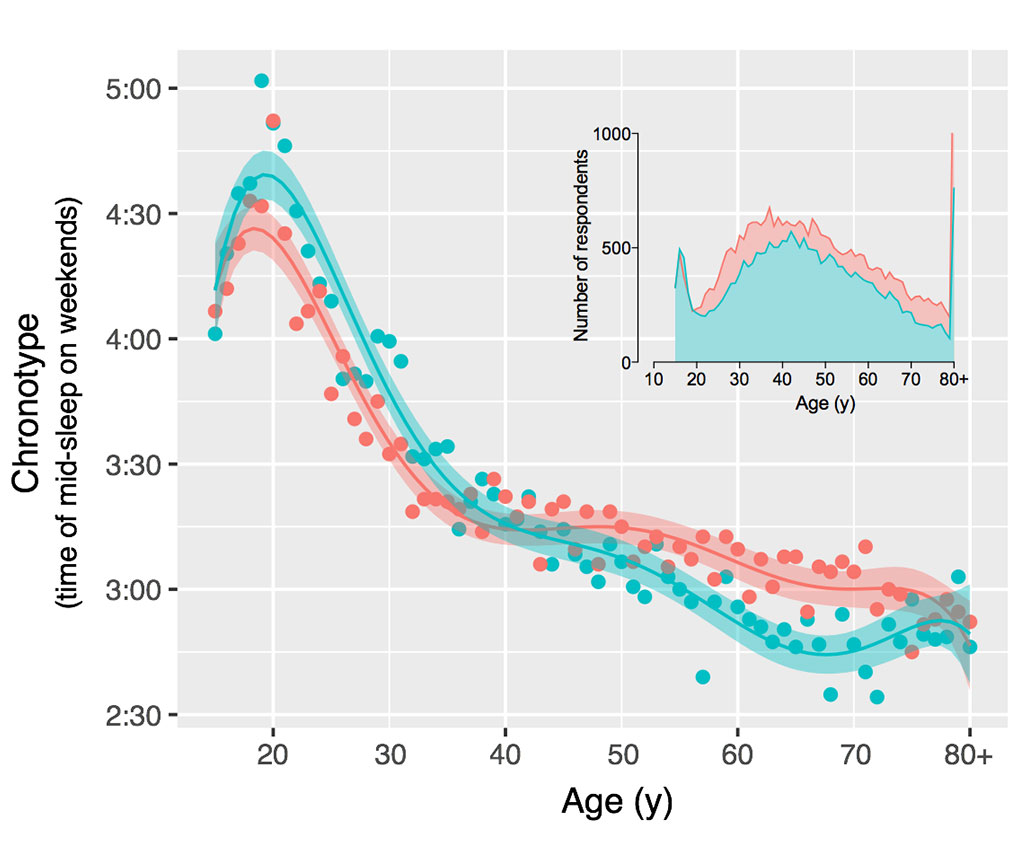
Roenneberg proposes adjusting work and school schedules to better match individuals’ internal clocks. His own research indicates chronotypes change over the lifetime and reach peak lateness during the teenage years. A 7:30 a.m. school start time is essentially the middle of the night for teenagers.
It’s also becoming clear that our internal clocks govern more than our sleep. Daily oscillations in metabolism and hormone secretion also determine optimal times for eating and exercising.
“The science is coming out to suggest, hey, timing matters, too,” says Knutson. “In the idea of a healthy lifestyle — with diet, exercise, and sleep — it isn’t just how much or what, but when as well.”
CONTENT PROVIDED BY
BrainFacts/SfN
Discussion Questions
1. How has access to electricity changed sleep patterns?
2. What role do genes play in circadian rhythms and chronotypes?
3. How does social jetlag hurt our health?
References
Beale, A. D., Pedrazzoli, M., Gonçalves, B. da S. B., Beijamini, F., Duarte, N. E., Egan, K. J., Knutson, K. L., Schantz, M. von, & Roden, L. C. (2017). Comparison between an African town and a neighbouring village shows delayed, but not decreased, sleep during the early stages of urbanisation. Scientific Reports, 7(1), 5697. https://doi.org/10.1038/s41598-017-05712-3
Feldman, J. F., & Hoyle, M. N. (1973). Isolation of circadian clock mutants of Neurospora crassa. Genetics, 75(4), 605–613.
Fischer, D., Lombardi, D. A., Marucci-Wellman, H., & Roenneberg, T. (2017). Chronotypes in the US – Influence of age and sex. PLOS ONE, 12(6), e0178782. https://doi.org/10.1371/journal.pone.0178782
Jones, C. R., Campbell, S. S., Zone, S. E., Cooper, F., DeSano, A., Murphy, P. J., Jones, B., Czajkowski, L., & Ptácek, L. J. (1999). Familial advanced sleep-phase syndrome: A short-period circadian rhythm variant in humans. Nature Medicine, 5(9), 1062–1065. https://doi.org/10.1038/12502
Jones, S. E., Lane, J. M., Wood, A. R., van Hees, V. T., Tyrrell, J., Beaumont, R. N., Jeffries, A. R., Dashti, H. S., Hillsdon, M., Ruth, K. S., Tuke, M. A., Yaghootkar, H., Sharp, S. A., Jie, Y., Thompson, W. D., Harrison, J. W., Dawes, A., Byrne, E. M., Tiemeier, H., … Weedon, M. N. (2019). Genome-wide association analyses of chronotype in 697,828 individuals provides insights into circadian rhythms. Nature Communications, 10(1), 343. https://doi.org/10.1038/s41467-018-08259-7
Kalmbach, D. A., Schneider, L. D., Cheung, J., Bertrand, S. J., Kariharan, T., Pack, A. I., & Gehrman, P. R. (2017). Genetic Basis of Chronotype in Humans: Insights From Three Landmark GWAS. Sleep, 40(2). https://doi.org/10.1093/sleep/zsw048
Katzenberg, D., Young, T., Finn, L., Lin, L., King, D. P., Takahashi, J. S., & Mignot, E. (1998). A CLOCK polymorphism associated with human diurnal preference. Sleep, 21(6), 569–576. https://doi.org/10.1093/sleep/21.6.569
Knutson, K. L., & Schantz, M. von. (2018). Associations between chronotype, morbidity and mortality in the UK Biobank cohort. Chronobiology International, 35(8), 1045–1053. https://doi.org/10.1080/07420528.2018.1454458
Knutson, K. L., Wu, D., Patel, S. R., Loredo, J. S., Redline, S., Cai, J., Gallo, L. C., Mossavar-Rahmani, Y., Ramos, A. R., Teng, Y., Daviglus, M. L., & Zee, P. C. (2017). Association Between Sleep Timing, Obesity, Diabetes: The Hispanic Community Health Study/Study of Latinos (HCHS/SOL) Cohort Study. Sleep, 40(4). https://doi.org/10.1093/sleep/zsx014
Konopka, R. J. (1979). Genetic dissection of the Drosophila circadian system. Federation Proceedings, 38(12), 2602–2605.
Konopka, R. J., & Benzer, S. (1971). Clock mutants of Drosophila melanogaster. Proceedings of the National Academy of Sciences of the United States of America, 68(9), 2112–2116. https://doi.org/10.1073/pnas.68.9.2112
Merikanto, I., Lahti, T., Puolijoki, H., Vanhala, M., Peltonen, M., Laatikainen, T., Vartiainen, E., Salomaa, V., Kronholm, E., & Partonen, T. (2013). Associations of chronotype and sleep with cardiovascular diseases and type 2 diabetes. Chronobiology International, 30(4), 470–477. https://doi.org/10.3109/07420528.2012.741171
Ralph, M. R., & Menaker, M. (1988). A mutation of the circadian system in golden hamsters. Science (New York, N.Y.), 241(4870), 1225–1227. https://doi.org/10.1126/science.3413487
Roenneberg, T., Kuehnle, T., Juda, M., Kantermann, T., Allebrandt, K., Gordijn, M., & Merrow, M. (2007). Epidemiology of the human circadian clock. Sleep Medicine Reviews, 11(6), 429–438. https://doi.org/10.1016/j.smrv.2007.07.005
Roenneberg, T., Pilz, L. K., Zerbini, G., & Winnebeck, E. C. (2019). Chronotype and Social Jetlag: A (Self-) Critical Review. Biology, 8(3). https://doi.org/10.3390/biology8030054
Roenneberg, T., Wirz-Justice, A., & Merrow, M. (2003). Life between clocks: Daily temporal patterns of human chronotypes. Journal of Biological Rhythms, 18(1), 80–90. https://doi.org/10.1177/0748730402239679
Toh, K. L., Jones, C. R., He, Y., Eide, E. J., Hinz, W. A., Virshup, D. M., Ptácek, L. J., & Fu, Y. H. (2001). An hPer2 phosphorylation site mutation in familial advanced sleep phase syndrome. Science (New York, N.Y.), 291(5506), 1040–1043. https://doi.org/10.1126/science.1057499
Vetter, C., Devore, E. E., Ramin, C. A., Speizer, F. E., Willett, W. C., & Schernhammer, E. S. (2015). Mismatch of Sleep and Work Timing and Risk of Type 2 Diabetes. Diabetes Care, 38(9), 1707–1713. https://doi.org/10.2337/dc15-0302
Vitaterna, M. H., King, D. P., Chang, A. M., Kornhauser, J. M., Lowrey, P. L., McDonald, J. D., Dove, W. F., Pinto, L. H., Turek, F. W., & Takahashi, J. S. (1994). Mutagenesis and mapping of a mouse gene, Clock, essential for circadian behavior. Science (New York, N.Y.), 264(5159), 719–725. https://doi.org/10.1126/science.8171325
Wittmann, M., Dinich, J., Merrow, M., & Roenneberg, T. (2006). Social jetlag: Misalignment of biological and social time. Chronobiology International, 23(1–2), 497–509. https://doi.org/10.1080/07420520500545979
Wright, K. P., McHill, A. W., Birks, B. R., Griffin, B. R., Rusterholz, T., & Chinoy, E. D. (2013). Entrainment of the Human Circadian Clock to the Natural Light-Dark Cycle. Current Biology, 23(16), 1554–1558. https://doi.org/10.1016/j.cub.2013.06.039
Yu, J. H., Yun, C.-H., Ahn, J. H., Suh, S., Cho, H. J., Lee, S. K., Yoo, H. J., Seo, J. A., Kim, S. G., Choi, K. M., Baik, S. H., Choi, D. S., Shin, C., & Kim, N. H. (2015). Evening chronotype is associated with metabolic disorders and body composition in middle-aged adults. The Journal of Clinical Endocrinology and Metabolism, 100(4), 1494–1502. https://doi.org/10.1210/jc.2014-3754

















