Light Molecules
- Published22 Mar 2008
- Reviewed13 Aug 2012
- Source BrainFacts/SfN
The discovery of light-activated molecules is enabling scientists to develop new tools to explore how specific types of nerve cells are interconnected and how they function in circuits in the brain. The new technology allows researchers to use light to study and even manipulate brain activity. Someday, these discoveries may result in improved and more precise therapies that target only diseased cells and avoid unwanted side effects in disorders such as Parkinson’s, depression, chronic pain, and epilepsy.
How algae move toward and away from light
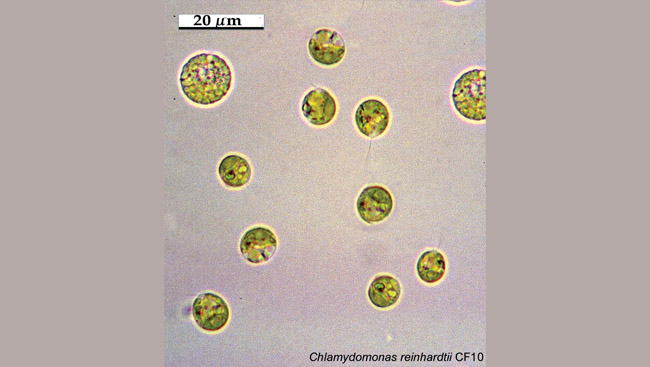
The study of obscure algae found in pond scum may not seem like the quickest path to curing neurological disorders like Parkinson’s disease. Yet green algae may hold the key to solving some of the most difficult problems in neuroscience.
A Scientific Puzzle
For centuries scientists have studied single-celled organisms such as green algae, puzzled by how they could sense and move toward and away from light without eyes. Only within the past few years did researchers discover that this movement was controlled by molecules called channelrhodopsins, photosensitive molecules analogous to those found in the retina of the human eye. Unlike molecules in the retina, however, these channelrhodopsins can generate electrical signals directly that allow this behavior in response to light.
An Unexpected Finding
Several independent research groups identified examples of these genes when they were combing through a database of the green algae genome and noticed the similarity between retinal pigment-containing proteins and these molecules. Further research suggested an amazing discovery: the new channelrhodopsins, in addition to sensing light, might also function as ion channels — proteins that allow charged particles to move into and out of cells and generate electrical signals. In the nervous system, ion channels are essential to transmit signals from one nerve cell to another.
The next exciting step was discovering that light can indeed open these channels directly and thereby let ions flow in sufficient quantities to excite brain cells, first tested simply in a dish in the laboratory. Researchers then discovered related molecules that can turn cells off: one extreme organism, salt-loving bacteria that grows in some desert lakes, has a light-activated ion channel “pump” that could be used to shut cells down.
Together, these two light-activated channels — the channelrhodopsin and the pump, one providing excitation and the second inhibition — provide exciting new tools for controlling the electrical activity of neurons.
Activating neurons in living animals with light
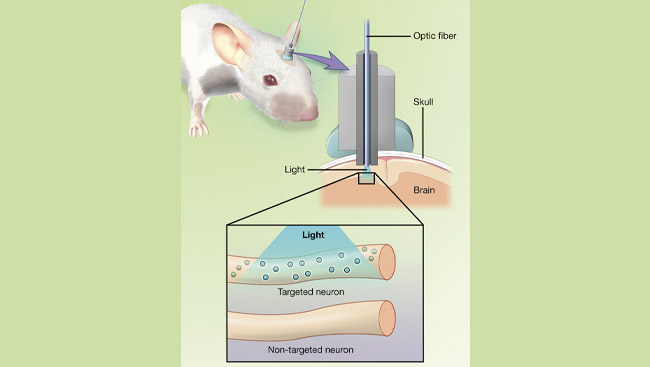
Shortly after the discovery of channelrhodopsins, it occurred to researchers that these new molecules could achieve a long-standing goal in neuroscience: a non-invasive way to turn neural activity on in particular parts of our brains.
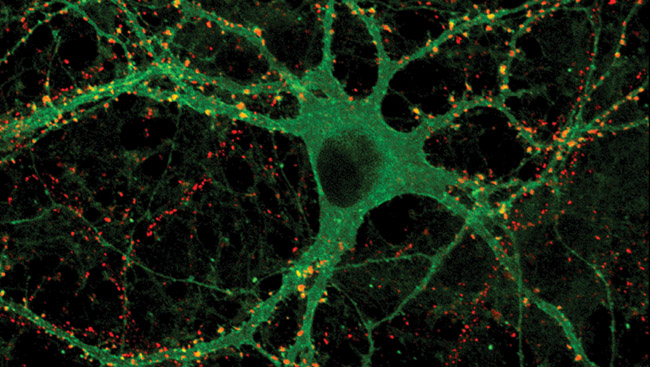
Using modern genetic methods, light-activated channels can be added to neurons that do not normally have them. This allows scientists to turn neurons in a living brain on or off simply by exposing them to light.
Developing Therapies and Probing Brain Function
Studies in worms and other animals show that neurons or muscles made to produce channelrhodopsins can be activated and deactivated quickly by light in living animals, eliciting behaviors that previously had been produced only by mechanical or electrical stimulation.
Research using light-activated channels in genetically-modified mice has also shown the potential for developing therapies and for probing the function of specific parts of the brain in mammals. For example, a study in some types of blind mice found that responses to light could be restored when channelrhodopsins were added to neurons in the inner retina. The findings show that light can be used to precisely manipulate neural activity in the living brain.
Light-activated channels also have the potential to advance our understanding of brain function and circuitry. Neuroscientists will be able to control the activity of specific groups of neurons remotely, with nothing more than pulses of light. By activating or inactivating specific groups of neurons, neuroscientists can understand how these neurons function in the brain to produce particular behaviors.
Better diagnosis and non-invasive techniques
With new technology, the light-activated channel discoveries can be used to map neural circuitry with extreme precision, improving diagnosis and offering new treatments.
In the future, the ability to activate or inactivate parts of the brain noninvasively has the potential to revolutionize the understanding of a number of neurological disorders, including those that arise because parts of the brain are either too active or not active enough — such as Parkinson’s disease in which cells that make dopamine are lost, causing altered activity levels in downstream circuits.
In the longer term, information gained about particular brain structures involved in behavior, emotion, and cognition is likely to help scientists design effective new treatment strategies for a number of brain disorders, not only for Parkinson’s disease but other neurological and psychiatric disorders including epilepsy and depression.
Non-Invasive Treatments, Fewer Side Effects
Non-invasive treatment with light could reduce problems with the current technologies for brain stimulation because the method is precise and fast. For instance, some Parkinson’s disease treatment involves the use of electrodes implanted into particular brain regions. However, sometimes these procedures can be ineffective, perhaps because the correct cell type is not targeted, and can cause serious side effects due to activation of surrounding brain areas not targeted for treatment.
Moreover, selective activation of targeted neurons would result in fewer side effects. Unlike electrical stimulation, this method could be used on genetically defined neuronal populations, which means that only the targeted neurons would be affected.
Another exciting potential application is to design neural prosthetic devices that use light to activate neurons that could take over the function of a limb. Although a number of technical challenges still must be solved before light-activated channels can be used effectively for these kinds of therapeutic strategies, the potential uses are numerous.
CONTENT PROVIDED BY
BrainFacts/SfN
Also In Archives
Trending
Popular articles on BrainFacts.org