Illuminating the Brain
- Published1 May 2012
- Reviewed1 May 2012
- Author Jennifer Carr
- Source BrainFacts/SfN
The discovery of a protein that gives jellyfish their colorful glow revolutionized scientists' view of the nervous system, allowing them to add color to what had only been seen in black and white.
With this protein and others identified in the wild, researchers are applying colors to brain cells that otherwise appear identical, enabling them to map where multiple cells begin and end, and the connections they make to each other. Understanding these connections and their susceptibility to change could help researchers better understand brain dysfunction in a variety of disorders, including autism, schizophrenia, Alzheimer's disease, and epilepsy.
A Glowing Jellyfish Protein Brightens Cells
More than 100 years ago, scientists got their first glimpse at brain cells under a microscope after successfully staining the cells in a single color. The technique made it possible for researchers to view entire brain cells with their long and branching processes for the first time; however, because the cells all appeared the same color and often overlapped it was difficult to tell one from the other. Only recently did researchers find a way to paint cells in a variety of hues — allowing them to distinguish individual cells and map the brain's connections.
A F of Lightlash
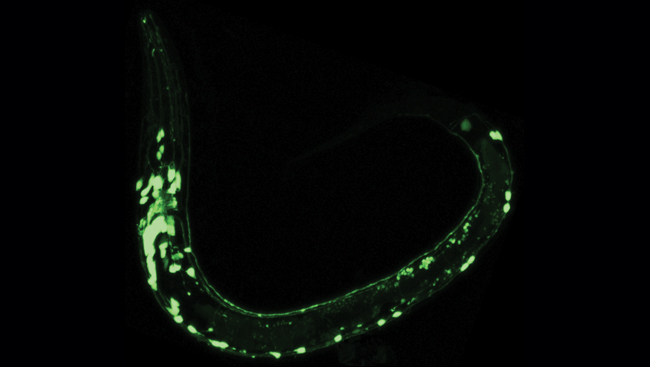
In the 1960s, researchers curious about the chemical reactions that enable some animals to emit light began studying the jellyfish Aequorea victoria, which glows green when agitated. They soon traced the colorful glow to green fluorescent protein (GFP), a molecule that emits green light when illuminated with blue or ultraviolet light.
In 1994, researchers began to unlock the potential for using GFP in biological research when they showed they could put it into the roundworm, Caenorhabditis elegans, and the fruit fly, Drosophila melanogaster — two major animal models in biology. Researchers soon discovered they could use GFP to illuminate the inner workings of cells. For example, it was used to light up nerve cells that respond to touch in C. elegans, permitting scientists to see these cells in different areas of the body and over various stages of development.
Creating a Wider Palette
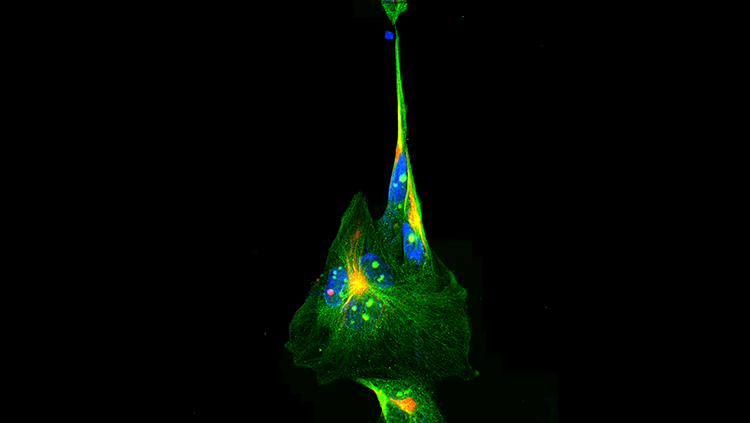
Proteins perform a flurry of activities to support the successful operation of each cell. In order to determine the role each one plays, researchers recognized the value of having more fluorescent color options than just green to parse out the many things occurring simultaneously inside a cell. Altering the structure of GFP so that it could absorb and emit light in other areas of the color spectrum produced different colors, such as blue and yellow. Researchers also identified and manipulated other fluorescent proteins found in the wild to make additional colors.
Scientists Martin Chalfie, Osamu Shimomura, and Roger Tsien shared the 2008 Nobel Prize in Chemistry for this research.
Animal research guides new ways to view brain
The human brain contains 100 billion nerve cells that snake and branch to form some 100 trillion connections, making the task of tracking all of the connections about as simple as trying to pinpoint the number of times strands of spaghetti touch in a bowl. Researchers who set out to map the much smaller mouse brain — home to 100 million nerve cells — soon recognized they needed a technique that would assign a different color to each cell.
Technicolor Brain Makes Pathways More Visible
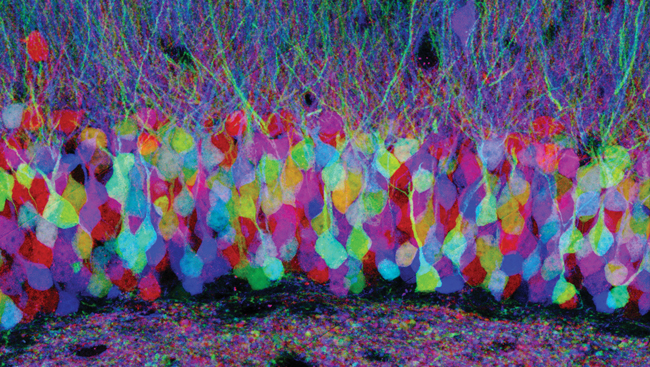
In 2007, researchers found a way to make each brain cell emit one of nearly 100 colors and distinguish neighboring cells. They genetically engineered mice to carry multiple copies of a chain of three or four genes for different colored fluorescent proteins. The mice were mated with others that made an enzyme that assigned one fluorescent protein per chain at random. In each cell, the combination of the colors emitted from each chain led to unique color blends. Just as a television produces a wide spectrum of colors by mixing red, green, and blue pixels, this so-called "brainbow" technique cast neighboring cells in an assortment of colors — from aquamarine to magenta.
With each cell a different color, brainbow enabled scientists to follow nerve cells as they forked and formed synapses, or junctions, with other cells in the brain. Brainbow has since been used to explore brain connection patterns in other lab animals, including fruit flies and zebrafish.
Researchers are using fluorescent proteins to improve other tools that track pathways in the brain as well. For example, one method takes advantage of the natural ability of some viruses to spread through the brain, cell by cell. By altering the viruses to contain GFP, researchers can track the connections between one brain cell and the next.
Neuroscience has benefited greatly from the identification of GFP and other fluorescent proteins. Scientists continue to use these proteins as tools to explore the brain in a variety of ways.
Pathways to Discovery
GFP is now used to track everything from how nerve cells communicate and cancer spreads through the body to the fate of stem cells and how HIV travels from infected to non-infected cells. GFP also has helped researchers explore the effects of gene mutations on the nervous system, discover the genes that help determine nerve cell number, and label individual cells for additional research.
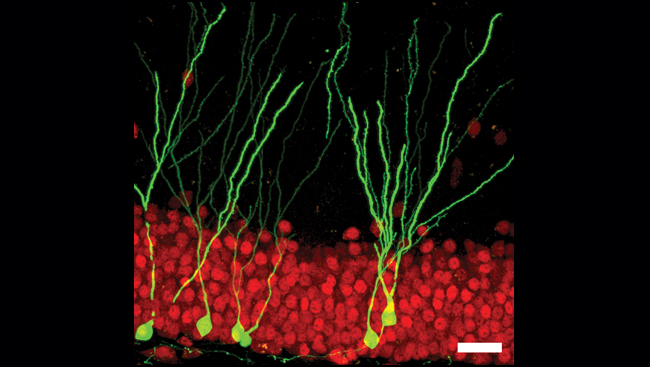
Neuroscientists are now using GFP to uncover the identities of individual nerve cells in mice — including the chemicals they use to communicate, the connections they make, and how such connections reorganize over time. Understanding patterns of brain connectivity and how they change during development may lead researchers to a better understanding about how the nervous system is built. Before researchers can begin to compile this information into meaningful maps of the brain, however, they must first tackle the challenge of storing immense amounts of data.
The Need for Bigger Computers
Researchers have completed a map of the C. elegans brain, which has 302 nerve cells — a tiny number compared to the 100 billion nerve cells in humans and 100 million in mice. Collecting and storing information about all nerve cells and their connections presents a formidable challenge for today's data technology. For instance, to store the images necessary to form a picture of 1 cubic millimeter of a mouse brain — about the size of the eye of a needle — requires roughly one petabyte of computer memory — the amount required to store more than 212,000 DVDs. Overcoming the challenge of storing such immense amounts of data will require collaboration across scientific disciplines and institutions, tapping into computational research underway at the National Science Foundation.
Examining the human brain on this scale, while challenging, is also a goal for scientists. To date, the National Institutes of Health has invested roughly $40 million to support the Human Connectome Project, which aims to one day map the circuitry of the healthy adult human brain. Such a map could revolutionize the diagnoses and treatments for diseases where faulty wiring is believed to play a role — such as autism and schizophrenia — and show how changes in wiring patterns contribute to brain dysfunction in aging.
CONTENT PROVIDED BY
BrainFacts/SfN