What the Brain of a 104-Year-Old Nun Taught Us About Vascular Dementia
- Published14 May 2020
- Author Alexis Wnuk
- Source BrainFacts/SfN
Over the last three decades, conditions damaging blood vessels, like obesity, high blood pressure, and high cholesterol, have also been linked to dementia and Alzheimer’s disease. The observations raise the intriguing possibility that vascular problems — rather than toxic proteins — kickstart the disease process.
A Neglected Piece of the Puzzle
Dressed in a black and white habit, 104-year-old Sister Matthia beams for the camera. Her wrinkled hands clasp a pair of knitting needles. A finished pink mitten sits in her lap. Three months before her death in December 1998, Sister Matthia remained quick-witted and sharp.
As a participant in the landmark Nun Study, Sister Matthia was one of 678 nuns at convents across the U.S. who volunteered to help epidemiologist David Snowdon study aging and Alzheimer’s disease. They agreed to complete a regular battery of cognitive and physical assessments, and most also donated their brains to the research effort after they died.

The work, which began at the University of Minnesota in 1986 and moved to the University of Kentucky before returning to Minnesota in 2009, revealed important lessons about healthy aging, including why some people, like Sister Matthia, are resilient to Alzheimer’s disease even though their brains displayed the telltale signs.
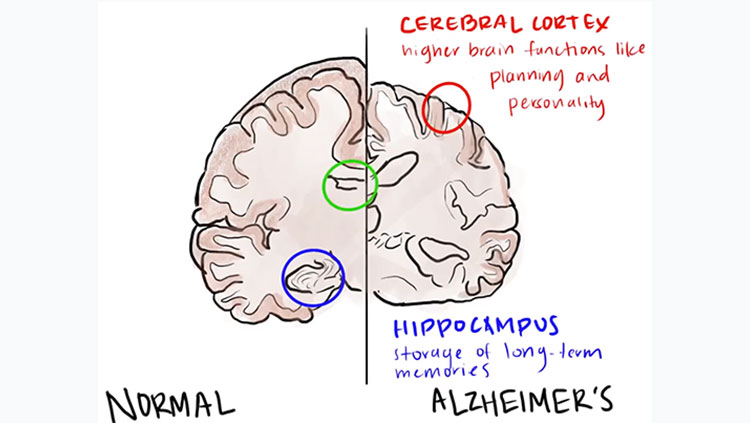
The Nun Study researchers unearthed their first clues in 1997 after examining the brains of 102 nuns who had died, each of whom had taken a battery of cognitive tests roughly one year prior to death. Of the 61 who possessed the sticky plaques and tangled proteins characteristic of Alzheimer’s disease, those whose brains also showed signs of stroke were far more likely to be demented. In other words, problems with the blood vessels supplying the brain compounded the damage wrought by Alzheimer’s.
Since then, the link between conditions that damage blood vessels, like obesity, high blood pressure, and high cholesterol, and cognitive decline and dementia, including Alzheimer’s disease, has only strengthened. Still, for the past 30 years, all efforts to combat Alzheimer’s have focused on the errant forms of two proteins: amyloid-beta and tau. Despite significant advances in our understanding of the disease, we’re no closer to a cure.
In order to make progress, scientists are looking anew at a neglected piece of the puzzle: damage to the remarkable web of blood vessels nourishing the brain.
The Possibility of Prevention
The pink, wrinkled mass of tissue that is the human brain inexplicably gives rise to memory, consciousness, and the human experience. Such effort makes it ravenous. A mere 2% of body mass, the human brain guzzles 20% of the body’s energy supply.
Charged with delivering oxygen and nutrients, a unique web of blood vessels covers the brain. Unlike any other blood vessels, these pack their cells together tightly, sealing off the brain from toxins and pathogens. They maintain consistent cerebral blood flow despite dips and surges in blood pressure. They sense changes in brain activity, rerouting blood flow where it’s needed most.
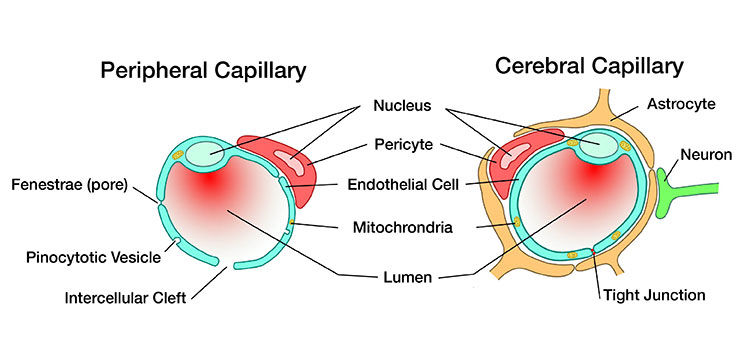
The system is vulnerable to slight perturbations. “Like anything complicated, sooner or later, something’s going to go wrong,” says Weill Cornell Medical College neurologist and neuroscientist Costantino Iadecola, who’s spent the last three decades studying the relationship between the brain and its blood vessels, and how it contributes to dementia.
When Alois Alzheimer discovered the signature plaques of presenile dementia in 1907, scientists thought dementia was caused by the hardening, stiffening, and narrowing of the arteries supplying the brain. Still, imaging experiments in the 1970s showed blood flow was relatively normal in older people with dementia.
In a 1974 research report, Western University neuroscientist Vladimir Hachinski suggested atherosclerosis instead damages the brain by causing strokes, each of which interrupts blood flow causing small patches of tissue to die. Depending on the number, size, and location of these necrotic areas — called infarcts — dementia may result. This proved a paradigm shift because unlike atherosclerosis stroke could be prevented. “He gave us hope that maybe by preventing stroke, we could prevent dementia,” Iadecola says.
But not Alzheimer’s disease because the working theory held that it was caused by the accumulation of toxic plaques. That supposition received significant boosts in the 1980s and 90s when separate research groups isolated and identified the misfolded protein in plaques as amyloid-beta, and pinpointed the genetic mutation responsible for inherited early-onset disease and the one responsible for late-onset sporadic disease.
A Spectrum of Dementias
Medical research often moves most rapidly when scientists target drugs to specific molecules and biological pathways. The discovery of genes and proteins accounting for Alzheimer’s disease from early onset to late onset raised significant hopes.
“The whole field shifted towards … Alzheimer’s disease to the point that nobody thought about vascular dementia as an important contribution anymore,” Iadecola says.
Even so, epidemiological studies linked high blood pressure, diabetes, and high levels of triglycerides with the development of dementia later in life. The Nun Study researchers found half of the dementia patients possessed amyloid plaques and infarcts in their brains.
In 1997, Iadecola looked at mice genetically engineered to overproduce amyloid-beta. The mice suffered impaired blood flow in their brains long before they accumulated plaques or cognitive problems.
What if vascular problems — and not plaques — kickstart the disease process?
In 2016, researchers at the Montreal Neurological Institute analyzed brain scans and blood and cerebrospinal fluid from more than 1,000 older adults. Part of the Alzheimer’s Disease Neuroimaging Initiative, which tracks the progression of cognitive impairment over time in order to identify early biomarkers for dementia, the researchers found vascular problems were the earliest and strongest predictors of late-onset Alzheimer’s. In 2019, researchers at the University of Southern California found cognitively impaired older adults had leakier blood-brain barriers.
Nearly one in three older adults possess Alzheimer’s characteristic plaques yet show none of the expected cognitive deficits. What’s more, estimates from several studies suggest mixed dementia — one marked by both plaques and vascular damage — may be the most common form of dementia. Iadecola sees dementia as a continuum: at one end resides the rare genetic disorder CADASIL that damages blood vessels and invariably leads to dementia. At the other, the inherited, early-onset form of Alzheimer’s disease where plaques accumulate and choke the brain. Fifty percent of people with dementia fall somewhere in the middle, Iadecola says.
“We need to train a generation of people who think neurons and vessels together,” Iadecola says. “Only by doing so are we going to be able to move forward in diseases like dementia, where the two are intimately related.”
CONTENT PROVIDED BY
BrainFacts/SfN
Discussion Questions
1) What clues to understanding Alzheimer’s disease were revealed with the Nun Study?
2) What caused researchers to shift their focus from plaques to blood flow in the investigations of dementia?
3) Why are vascular health issues a predictor of dementia for older adults?
References
Bowler, J. V. (2007). Modern concept of vascular cognitive impairment. British Medical Bulletin, 83, 291–305. doi: 10.1093/bmb/ldm021
de la Torre, J. C. (2002). Alzheimer disease as a vascular disorder: Nosological evidence. Stroke, 33(4), 1152–1162. doi: 10.1161/01.str.0000014421.15948.67
Gottesman, R. F., Schneider, A. L. C., Zhou, Y., Coresh, J., Green, E., Gupta, N., … Mosley, T. H. (2017). Association Between Midlife Vascular Risk Factors and Estimated Brain Amyloid Deposition. JAMA, 317(14), 1443–1450. doi: 10.1001/jama.2017.3090
Hachinski, V. C., Lassen, N. A., & Marshall, J. (1974). Multi-infarct dementia. A cause of mental deterioration in the elderly. Lancet (London, England), 2(7874), 207–210. doi: 10.1016/s0140-6736(74)91496-2
Hachinski, Vladimir C. (1991). Multi-Infarct Dementia: A Reappraisal. Alzheimer Disease & Associated Disorders, 5(2), 64–68. Retrieved from: https://journals.lww.com/alzheimerjournal/Abstract/1991/00520/Multi_Infarct_Dementia__A_Reappraisal.2.aspx
Iadecola, C. (2013). The pathobiology of vascular dementia. Neuron, 80(4), 844–866. doi: 10.1016/j.neuron.2013.10.008
Iadecola, C. (2017). The Neurovascular Unit Coming of Age: A Journey through Neurovascular Coupling in Health and Disease. Neuron, 96(1), 17–42. doi: 10.1016/j.neuron.2017.07.030
Iadecola, C., Duering, M., Hachinski, V., Joutel, A., Pendlebury, S. T., Schneider, J. A., & Dichgans, M. (2019). Vascular Cognitive Impairment and Dementia: JACC Scientific Expert Panel. Journal of the American College of Cardiology, 73(25), 3326–3344. doi: 10.1016/j.jacc.2019.04.034
Iturria-Medina, Y., Sotero, R. C., Toussaint, P. J., Mateos-Pérez, J. M., & Evans, A. C. (2016). Early role of vascular dysregulation on late-onset Alzheimer’s disease based on multifactorial data-driven analysis. Nature Communications, 7(1), 1–14. doi: 10.1038/ncomms11934
Kivipelto, M., Ngandu, T., Fratiglioni, L., Viitanen, M., Kåreholt, I., Winblad, B., … Nissinen, A. (2005). Obesity and Vascular Risk Factors at Midlife and the Risk of Dementia and Alzheimer Disease. Archives of Neurology, 62(10), 1556–1560. doi: 10.1001/archneur.62.10.1556;
Lemonick, M. D., & Mankato, A. P. (2001, May 14). The Nun Study: How one scientist and 678 sisters are helping unlock the secrets of Alzheimer’s. Time. Retrieved from http://content.time.com/time/world/article/0,8599,2047984,00.html
Nasrallah, I. M., Pajewski, N. M., Auchus, A. P., Chelune, G., Cheung, A. K., Cleveland, M. L., … Bryan, R. N. (2019). Association of Intensive vs Standard Blood Pressure Control With Cerebral White Matter Lesions. JAMA, 322(6), 524–534. doi: 10.1001/jama.2019.10551
Nation, D. A., Sweeney, M. D., Montagne, A., Sagare, A. P., D’Orazio, L. M., Pachicano, M., … Zlokovic, B. V. (2019). Blood–brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nature Medicine, 25(2), 270–276. doi: 10.1038/s41591-018-0297-y
Niwa, K., Kazama, K., Younkin, S. G., Carlson, G. A., & Iadecola, C. (2002). Alterations in cerebral blood flow and glucose utilization in mice overexpressing the amyloid precursor protein. Neurobiology of Disease, 9(1), 61–68. doi: 10.1006/nbdi.2001.0460
Pajewski, N. M., Berlowitz, D. R., Bress, A. P., Callahan, K. E., Cheung, A. K., Fine, L. J., … Williamson, J. D. (2019). Intensive vs Standard Blood Pressure Control in Adults 80 Years or Older: A Secondary Analysis of the Systolic Blood Pressure Intervention Trial. Journal of the American Geriatrics Society. doi: 10.1111/jgs.16272
Pase, M. P., Satizabal, C. L., & Seshadri, S. (2017). Role of Improved Vascular Health in the Declining Incidence of Dementia. Stroke, 48(7), 2013–2020. doi: 10.1161/STROKEAHA.117.013369
Sierra, C. (2020). Hypertension and the Risk of Dementia. Frontiers in Cardiovascular Medicine, 7, 5. doi: 10.3389/fcvm.2020.00005
Snowdon, D. A. (1997). Aging and Alzheimer’s disease: Lessons from the Nun Study. The Gerontologist, 37(2), 150–156. doi: 10.1093/geront/37.2.150
Snowdon, David A., & Nun Study. (2003). Healthy aging and dementia: Findings from the Nun Study. Annals of Internal Medicine, 139(5 Pt 2), 450–454. doi: 10.7326/0003-4819-139-5_part_2-200309021-00014
Snowdon, David A., Tully, C. L., Smith, C. D., Riley, K. P., & Markesbery, W. R. (2000). Serum folate and the severity of atrophy of the neocortex in Alzheimer disease: Findings from the Nun Study. The American Journal of Clinical Nutrition, 71(4), 993–998. doi: 10.1093/ajcn/71.4.993
Sweeney, M. D., Montagne, A., Sagare, A. P., Nation, D. A., Schneider, L. S., Chui, H. C., … Zlokovic, B. V. (2019). Vascular dysfunction-The disregarded partner of Alzheimer’s disease. Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, 15(1), 158–167. doi: 10.1016/j.jalz.2018.07.222
Tyas, S. L., Snowdon, D. A., Desrosiers, M. F., Riley, K. P., & Markesbery, W. R. (2007). Healthy ageing in the Nun Study: Definition and neuropathologic correlates. Age and Ageing, 36(6), 650–655. doi: 10.1093/ageing/afm120
Williamson, J. D., Pajewski, N. M., Auchus, A. P., Bryan, R. N., Chelune, G., Cheung, A. K., … Wright, C. B. (2019). Effect of Intensive vs Standard Blood Pressure Control on Probable Dementia: A Randomized Clinical Trial. JAMA, 321(6), 553–561. doi: 10.1001/jama.2018.21442
Zhang, F., Eckman, C., Younkin, S., Hsiao, K. K., & Iadecola, C. (1997). Increased susceptibility to ischemic brain damage in transgenic mice overexpressing the amyloid precursor protein. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 17(20), 7655–7661. Retrieved from: https://www.ncbi.nlm.nih.gov/pubmed/9315887





.jpg)