Smell: An Overview
- Published28 Dec 2016
- Reviewed28 Dec 2016
- Author Stephani Sutherland
- Source BrainFacts/SfN
The discovery of a large family of receptors detecting odor solved the mystery of how our brains perceive smell.
Smell evokes powerful memories. Yet, it remained the most enigmatic of our senses until researchers discovered a large family of proteins that detect odors. Understanding these proteins has revealed the primal nature of smell, opened a window on neurodegenerative disorders, and unveiled a class of chemical sensors throughout the body.
The Discovery
The aroma of freshly baked cookies or the scent of a rose can evoke intense memories. For animals, the sense of smell exerts a powerful influence over behaviors including eating, mating, and avoiding danger. Despite its primal nature, the sense of smell remained an enigma until very recently.
Our senses allow us to detect physical changes around us and perceive the nature of our world. For sight that begins with the retina detecting light, but actually seeing a car speeding towards us requires the brain to receive the signals from the retina and to process it quickly. Perceiving that car as green is the result of the brain processing signals from sensory cells in the eye capable of detecting just three primary colors.
Centuries of experimentation culminating in a 1965 experiment demonstrated the role of “cone” cells in color vision. As a result, scientists postulated that, similar to how we see, smell-detecting olfactory neurons might pick up a few “primary” odors that the brain then turned into the vast number of odors we can detect.
It took a technological breakthrough to prove that assumption wrong. In the late 1980s, polymerase chain reaction (PCR) revolutionized molecular biology by providing a way to replicate small amounts of genetic material in order to study them. Richard Axel and Linda Buck employed PCR to hunt for odorant receptors.
They reasoned odorant receptors might belong to a large, diverse family of receptor proteins found in other tissues in the body, but they looked for such proteins only in the sensory neurons in the nose. Axel and Buck scanned the genetic material contained in rats’ olfactory neurons looking for sequences similar to those encoding the receptor family. In 1991, they isolated 18 unique receptor genes that were members of this family but were found only in olfactory sensory neurons. Those 18 were the first of what turned out to be more than 1000 genes in rodents encoding smell sensors, more commonly referred to today as olfactory receptors. For their work, Axel and Buck were awarded the 2004 Nobel Prize in Physiology or Medicine.
Deeper Knowledge
With olfactory receptors in hand, researchers around the world began the work of understanding how we can discern the difference between remarkably similar odor molecules. By 2002, they had established that the receptors represented a “super family” of more than 1000 individual proteins. Still, those numbers couldn’t account for all the smells in our world if each receptor detected only one particular odor.
By 1999, the scientific community knew that each receptor could in fact . That ability greatly increased the number of detectable odors, but not enough to fully account for our ability to smell many odors.
The discovery that individual receptors can respond to many different molecules was the key to understanding how a limited number of receptors allows us to detect and discern a vast number of odors. What’s more, any given odor molecule activates multiple olfactory receptors.
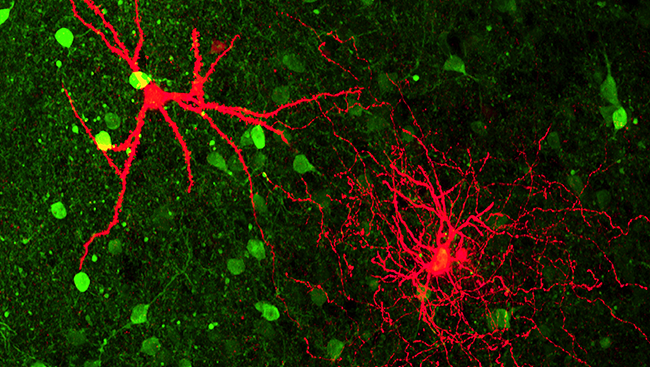
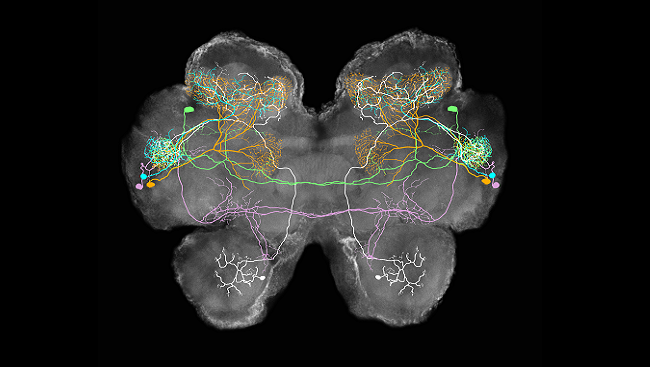
Perceiving smell begins with olfactory receptors in the nose and ends in the brain. Each smell activates a specific combination of olfactory neurons, which the brain decodes as a particular aroma. This “combinatorial” coding allows us to detect many more smells than we have specific receptors. Each chemical odorant triggers its own unique pattern of neural activity, leading to our perception of a particular smell.
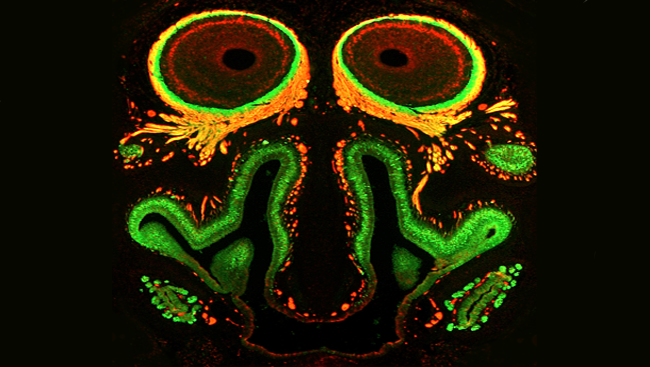
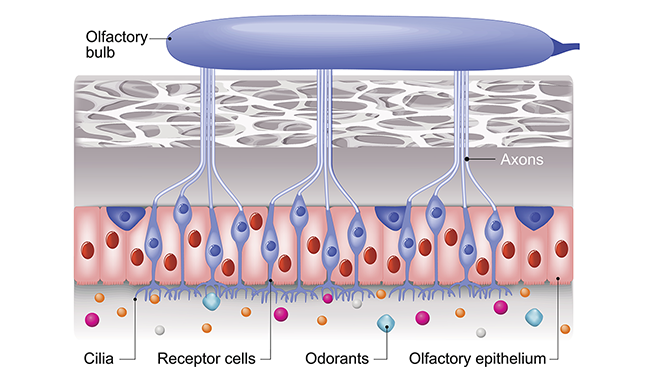
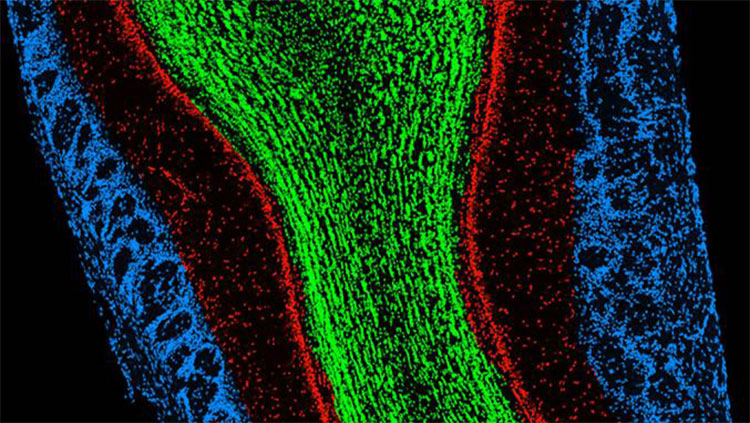
Buck, Axel, and others went on to study the organization of sensory neurons in the olfactory epithelium — the nervous tissue inside the nose — which helped describe how information flows from nose to brain. They described how those sensory neurons communicate with their targets — neurons in a brain structure called the olfactory bulb — and how information is passed along to higher relay stations, like the olfactory cortex.
What’s more, smell signals radiate to deeper regions of the brain, including the amygdala and hippocampus, areas critical for emotion and memory — one reason smells can evoke such powerful feelings.
Health Implications
The moment we catch a whiff of garlic sizzling in a pan, olfactory neurons in our noses spring into action sending electrical signals on a journey through our brains. The first stop is the olfactory centers where they transform into what we perceive as smell. As the signals travel to deeper regions of the brain, they can trigger emotion and memories as they arrive in the amygdala and hippocampus.
Although one could survive in the modern world without a sense of smell, losing it can signal something more pernicious in the brain. Patients with some neurodegenerative disorders including Alzheimer’s disease and Parkinson’s disease frequently struggle to detect and identify smells. These problems crop up long before their memories become impaired or they develop tremors. Studies indicate deficits in smell amongst healthy older adults can predict later cognitive decline or Parkinson’s disease.
Researchers continue to explore why this happens in order to develop early diagnostics and potentially new therapies aimed at the underlying cause of the damage.
Although the vast majority of olfactory receptors exist only in the nose, scientists have found some receptors in other tissues including the lungs, kidneys, skin, heart, muscle, colon, and brain. As a result, scientists are beginning to posit that they serve not just as smell receptors but as more general chemical sensors. Sperm, for example, use input from olfactory receptors to navigate to unfertilized eggs. In kidneys, they help mice regulate blood pressure. Lungs use olfactory receptors to detect noxious chemicals and constrict airways.
Researchers have also begun exploring how olfactory receptors throughout the body may possibly facilitate the effects of certain drugs.
The discovery of olfactory receptors opened a window on our sense of smell potentially leading to a better understanding of chemical signaling throughout the body. Through continued research funded by public and private institutions, scientists may find new roles for olfactory receptors as well as new opportunities to diagnose and treat neurodegenerative diseases like Alzheimer’s and Parkinson’s disease.
CONTENT PROVIDED BY
BrainFacts/SfN
References
Buck L, Axel R. A novel multigene family may encode odorant receptors: a molecular basis for odor recognition. Cell. Vol. 65, 175-18 (1991).
Buck L. The search for odorant receptors. Cell. 116(2 Suppl):S117-9 (2004).
Doty R. Smell and the degenerating brain. The Scientist. October 1, 2013.
Ho J, Perez-Aguilar JM, Gao L, Saven JG, Matsunami H,. Molecular recognition of ketamine by a subset of olfactory G protein–coupled receptors. Science Signaling. 8(370): ra33 (2015).
Malnic B, Hirono J, Sato T, Buck LB. Combinatorial receptor codes for odors. Cell. 96(5):713-23 (1999).
Nara K, Saraiva LR, Ye X, Buck LB. A large-scale analysis of odor coding in the olfactory epithelium. Journal of Neuroscience. 31(25):9179-91 (2011).
Zhang X, Firestein S. The olfactory receptor gene superfamily of the mouse. Nature Neuroscience. 5(2):124-33 (2002).